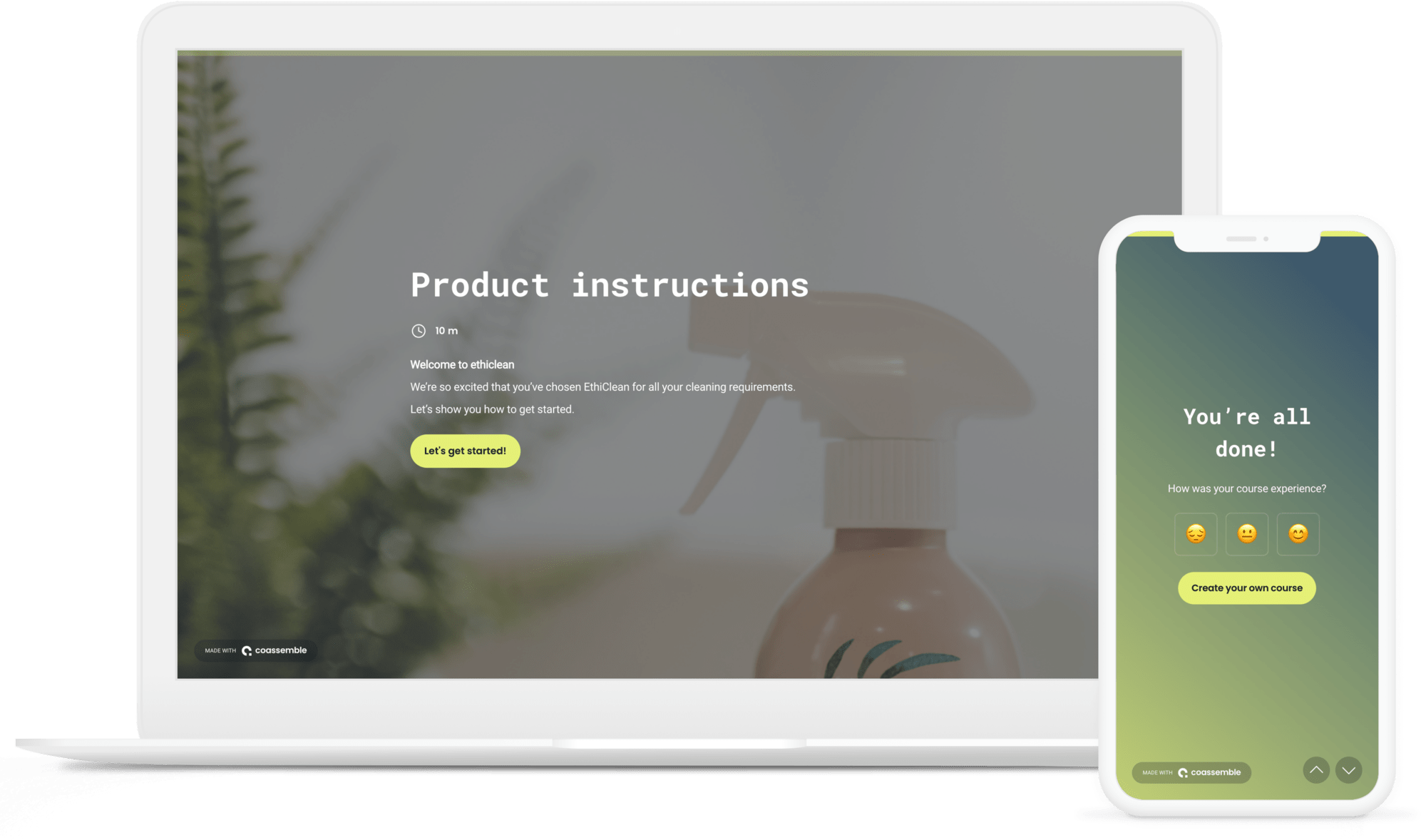
Device Instructions Template
Device Instructions Template - You can build an app. See for example guidance on medical. The fda has issued guidance documents on labelling and how to write instructions for users according to best practices. We’ve already helped hundreds of companies with their mdr compliance. Customize and download this smart device instruction manual. For every item or product, there must be an instruction. Capacity, capabilities and the functions of the product.
The fda has issued guidance documents on labelling and how to write instructions for users according to best practices. Features of the product in detail. These user manuals will be very useful for the buyers to know about how to use the products so that they won’t face any kind of incovenience. Unsure how to get started and how to get your eu mdr medical device certified?
The fda has issued guidance documents on labelling and how to write instructions for users according to best practices. Here are the instruction manual templates that you can download and use as your guidelines. Enhance this design & content with free ai. See for example guidance on medical. These user manuals will be very useful for the buyers to know about how to use the products so that they won’t face any kind of incovenience. In order to be effective and useful, an instruction or operation manual template should include the following important information:
Features of the product in detail. The fda has issued guidance documents on labelling and how to write instructions for users according to best practices. These user manuals will be very useful for the buyers to know about how to use the products so that they won’t face any kind of incovenience. Unsure how to get started and how to get your eu mdr medical device certified? Capacity, capabilities and the functions of the product.
Within our mydevices console, you have the ability to create device templates and codecs for various devices, such as sensors, actuators, gateways, tags, and more. The correct model number of the product. These user manuals will be very useful for the buyers to know about how to use the products so that they won’t face any kind of incovenience. In order to be effective and useful, an instruction or operation manual template should include the following important information:
Enhance This Design & Content With Free Ai.
The correct model number of the product. In order to be effective and useful, an instruction or operation manual template should include the following important information: The fda has issued guidance documents on labelling and how to write instructions for users according to best practices. Step wise guidelines for installation of the product.
You Can Build An App.
Here are the instruction manual templates that you can download and use as your guidelines. For every item or product, there must be an instruction. These user manuals will be very useful for the buyers to know about how to use the products so that they won’t face any kind of incovenience. Smart device instruction manual is in editable, printable format.
Capacity, Capabilities And The Functions Of The Product.
Complete name of the product. We’ve already helped hundreds of companies with their mdr compliance. Features of the product in detail. Unsure how to get started and how to get your eu mdr medical device certified?
When Some Product Is Being Manufactured, Then You Generally Have To Make The User Manuals As Well.
Customize and download this smart device instruction manual. See for example guidance on medical. Within our mydevices console, you have the ability to create device templates and codecs for various devices, such as sensors, actuators, gateways, tags, and more.
The correct model number of the product. The fda has issued guidance documents on labelling and how to write instructions for users according to best practices. Smart device instruction manual is in editable, printable format. Here are the instruction manual templates that you can download and use as your guidelines. We’ve already helped hundreds of companies with their mdr compliance.